Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
BioViotica
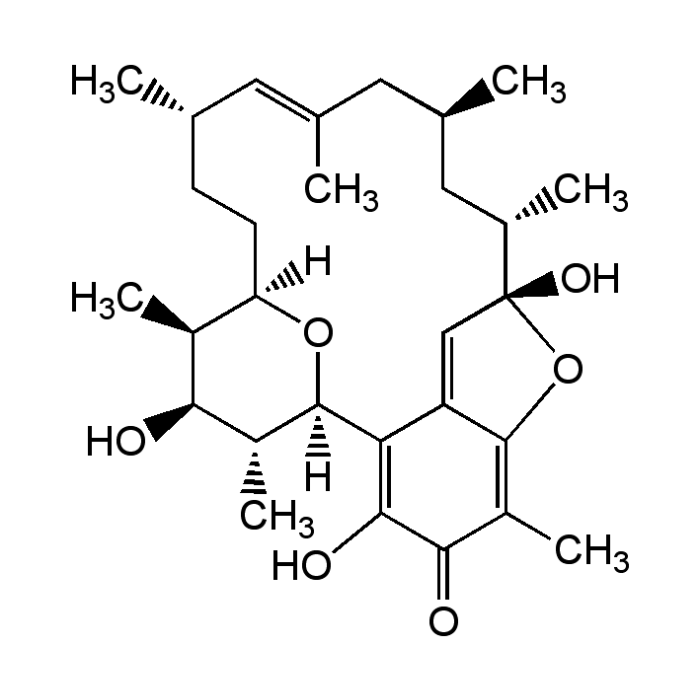
Kendomycin
As low as
95
CHF
CHF 95.00
In stock
Only %1 left
BVT-0001-C100100 µgCHF 95.00
BVT-0001-C500500 µgCHF 215.00
BVT-0001-MM252.5 mgCHF 580.00
| Product Details | |
|---|---|
| Synonyms | (-)-TAN 2162 |
| Product Type | Chemical |
| Properties | |
| Formula |
C29H42O6 |
| MW | 486.6 |
| CAS | 183202-73-5 |
| Source/Host Chemicals | Isolated from Streptomyces violaceoruber. |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Yellow powder. |
| Solubility | Slightly soluble in methanol or DMSO. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by BioViotica. |
| InChi Key | HKLDUJXJTQJSEJ-OLXNOMCWSA-N |
| Smiles | [H][C@]12CC[C@H](C)\C=C(C)\C[C@@H](C)C[C@H](C)[C@]3(O)OC4=C(C)C(=O)C(O)=C(C4=C3)[C@]([H])(O1)[C@H](C)[C@@H](O)[C@H]2C |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep under inert gas. Protect from light and oxygen. |
| Use/Stability |
Stable for at least 1 year after receipt when stored at -20°C. After reconstitution protect from light at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Antibiotic.
- Potent endothelin receptor antagonist.
- Anti-osteoporotic.
- Antibacterial.
- Anticancer agent. Cytotoxic against several carcinoma cell lines (GI50<100nM).
- 20S proteasome inhibitor. Induces apoptosis by inhibition of chymotrypsin-like proteasome activity.
Product References
- Structure and biosynthesis of kendomycin, a carboxylic ansa-compound from Streptomyces: H.B. Bode & A. Zeeck; J. Chem. Soc. Perkin Trans 1, 323 and 2665 (2000)
- Evidence for the mode of action of the highly cytotoxic Streptomyces polyketide kendomycin: Y.A. Elnakady, et al.; ChemBioChem. 8, 1261 (2007)
- Formal Synthesis of (−)-Kendomycin Featuring a Prins-Cyclization To Construct the Macrocycle: K.B. Bahnck & S.D. Rychnovsky; JACS 130, 13177 (2008)
- Interactions of the natural product kendomycin and the 20S proteasome: P. Beck, et al.; J. Mol. Biol. 426, 3108 (2014)