Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
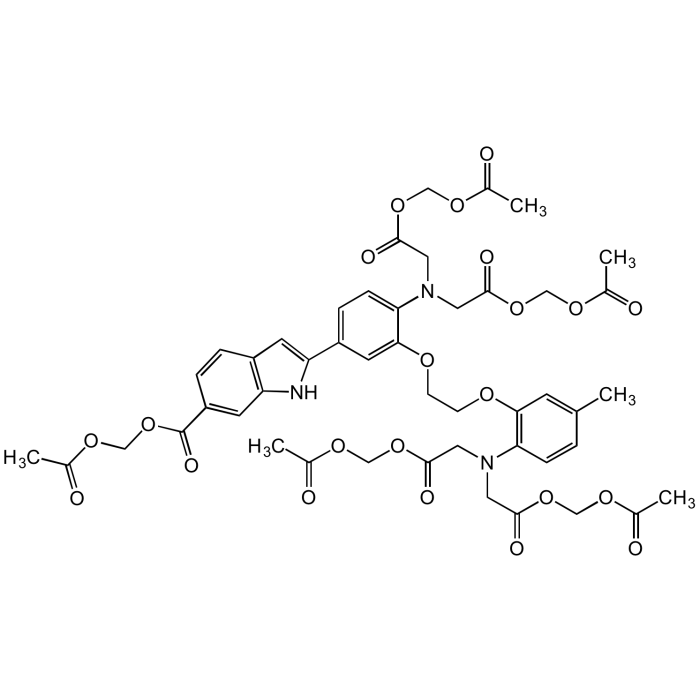
Indo 1-AM
| Product Details | |
|---|---|
| Synonyms | Indo-1-(acetoxymethyl) ester; 4-(6-Carboxy-2-indolyl)-4'-methyl-2,2'-(ethylenedioxy)dianiline-N,N,N',N'-tetraacetic acid tetrakis(acetoxymethyl) ester |
| Product Type | Chemical |
| Properties | |
| Formula | C47H51N3O22 |
| MW | 1009.93 |
| CAS | 112926-02-0 |
| Source/Host Chemicals | Synthetic. |
| Purity Chemicals | ≥90% (HPLC) |
| Appearance | Off-white solid. |
| Solubility | Soluble in DMSO or methanol. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | CAWBRCOBJNWRLK-UHFFFAOYSA-N |
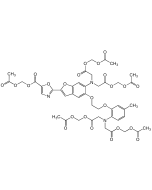
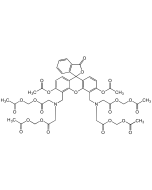
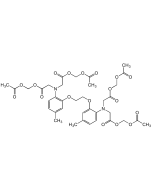
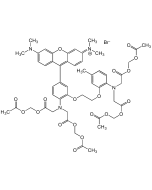
| Smiles | CC1=CC(OCCOC2=CC(C3=CC(C=CC(C(OCOC(C)=O)=O)=C4)=C4N3)=CC=C2N(CC(OCOC(C)=O)=O)CC(OCOC(C)=O)=O)=C(N(CC(OCOC(C)=O)=O)CC(OCOC(C)=O)=O)C=C1 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | -20°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
INDO 1-AM is a popular non-invasive UV-excitable calcium indicator. It is the cell-permeable ester derivative of INDO 1. After crossing the cell membrane, INDO 1-AM is rapidly hydrolyzed by cytoplasmic esterases to produce the ratiometric fluorescent calcium indicator INDO 1, which remains trapped within the cell. INDO 1/AM has been used to selectively monitor Ca2+ levels in mitochondria and in the cytosol. INDO 1 is ideal for analyses using flow cytometry, as it uses a single excitation source (usually the 351-364nm spectral lines of the argon-ion laser). In contrast to FURA 1, INDO 1 has a dual emission peak. The emission of INDO 1 shifts to 400nm (when bound to Ca2+) from 480nm in Ca2+-free environments. INDO 1 is prone to photobleaching, which limits its usefulness in methods involving microscopy. Calcium measurement is critical for numerous biological investigations. Fluorescent probes that show spectral responses upon binding Ca2+ have enabled researchers to investigate changes in intracellular free Ca2+ concentrations by using fluorescence microscopy, flow cytometry, fluorescence spectroscopy and fluorescence microplate readers. Spectral Data: λex 330nm; λem 450nm (with Calcium), λex 356nm; λem 478nm in methanol.
(1) G. Grynkiewicz, et al.; J. Biol. Chem. 260, 3440 (1985) | (2) M. Lopez, et al.; Cytometry 10, 165 (1989) | (3) J.P. Grierson, et al.; J. Neurophysiol. 67, 704 (1992) | (4) E.J. Griffiths, et al.; Am. J. Physiol. 273, C37 (1997) | (5) R.M. Paredes, et al.; Meth. A Comp. Meth. Enzymol. 46, 143 (2008) | (6) Z. Zhou, et al.; J. Physiol. 507, 379 (1998) | (7) S. Bailey & P.J. Macardle; J. Immunol. Meth. 311, 220 (2006) | (8) A. Nelemans; Methods Mol. Biol. 312, 47 (2006) | (9) R.W. Sabnis; Handbook of biological dyes and stains (2010)