Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
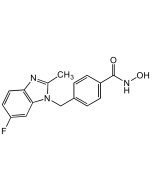
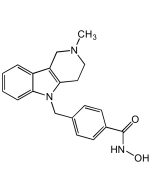
MBIMPH F-Analog 1 . hydrochloride
As low as
200
CHF
CHF 200.00
In stock
Only %1 left
AG-CR1-3908-M0011 mgCHF 200.00
AG-CR1-3908-M0055 mgCHF 800.00

| Product Details | |
|---|---|
| Synonyms | MBIMPH Fluorinated Analog 1; 3-Fluoro-N-hydroxy-4-((2-methyl-1H-benzo[d]imidazol-1-yl)methyl)benzamide |
| Product Type | Chemical |
| Properties | |
| Formula |
C16H14FN3O2 . HCl |
| MW | 299.3 . 36.5 |
| Purity Chemicals | ≥95% (HPLC) |
| Appearance | Off-white solid. |
| Solubility | Soluble in DMSO, ethanol, methanol or water. |
| Identity | Determined by 1H-NMR. |
| InChi Key | NWMOSGPUNITEFR-UHFFFAOYSA-N |
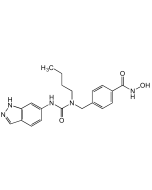
| Smiles | Cl.CC1=NC2=C(C=CC=C2)N1CC1=C(F)C=C(C=C1)C(=O)NO |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Cell permeable, potent and selective class IIb HDAC6 inhibitor (IC50 =3nM). Displays high selectivity over all other HDACs (IC50=0.03-20µM).
- Induces cellular α-tubulin, but not histone H3 hyperacetylation in Neuro-2a cells. Promotes mitochondrial transport.
- Shows improved kinetics and biochemical potency against HDAC6 compared to tubastatin A (AG-CR1-3900).
- HDAC6 deacetylates tubulin, HSP90 and the core histones (H2A, H2B, H3, H4). Histone deacetylases act via the formation of large multiprotein complexes. HDAC6 plays an important role in microtubule-dependent cell motility, transcriptional regulation, degradation of misfolded proteins and cell cycle and is involved in autophagy, inflammation, cancer and neurodegeneration.
Product References
- Bicyclic-capped histone deacetylase 6 inhibitors with improved activity in a model of axonal charcot-marie-tooth disease: S. Shen, et al.; ACS Chemical Neuroscience (submitted) (2015)