Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
anti-GBP1 (human), pAb (IN111)

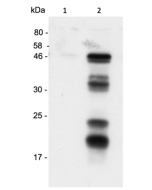
Method: GBP1 is analyzed by Western blot in cell extracts of HeLa cells treated overnight with 0, 10 or 100 ng/ml of IFN-γ (human) (rec.) (His) (Prod. No. CHI-HR-200IFNG). Cell extracts (20µl) are separated by SDS-PAGE under reducing conditions, transferred to nitrocellulose and incubated with anti-GBP1 (human), pAb (IN111) (2µg/ml). Proteins are visualized by TMB.
| Product Details | |
|---|---|
| Synonyms | Guanylate-binding Protein 1; GTP-binding Protein 1; Guanine Nucleotide-binding Protein 1; Interferon-induced Guanylate-binding Protein 1 |
| Product Type | Polyclonal Antibody |
| Properties | |
| Source/Host | Rabbit |
| Immunogen/Antigen | Recombinant human GBP1 protein (aa 200-592). |
| Application |
Western Blot: (1:500)* *Note: We recommend starting a dilution of 1:500 and to optimize the dilution depending on your cells/tissues. |
| Crossreactivity | Human |
| Specificity |
Recognizes endogenous human GBP1. |
| Purity | ≥95% (SDS-PAGE) |
| Purity Detail | Protein A affinity purified. |
| Concentration | 1mg/ml |
| Formulation | Liquid. In PBS containing 0.05% sodium azide. |
| Isotype Negative Control | |
| Other Product Data |
UniProt link P32455: GBP1 (human) |
| Accession Number | P32455 |
| Shipping and Handling | |
| Shipping | BLUE ICE |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
After opening, prepare aliquots and store at -20°C. Avoid freeze/thaw cycles. |
| Use/Stability | Stable for at least 1 year after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Guanylate-binding proteins (GBPs) are a family of antimicrobial proteins that are expressed by host cells in response to pathogens. Expression of GBPs is induced by both type I IFNs and IFN-γ. During infection with pathogen (S. Typhimurium or Salmonella), GBP1 binds first to surface of the bacteria, leading to the recruitment of GBP2, GBP3 and GBP4. GBP1 binds to the component LPS (Lipopolysaccharide) of the bacteria as a first step bringing caspase-4 to the surface of bacteria, leading to caspase-1 activation, Gasdermin D cleavage and finally pyroptotic cell death. GBP1 is cleaved and inactivated by Caspase-1. GBP1 also promotes Toxoplasma DNA detection by AIM2-ASC-Caspase-8 leading to apoptosis. GBP1 acts as a gatekeeper of cell death pathways, which respond specifically to infecting microbes.