Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
Chemodex
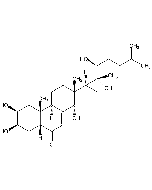
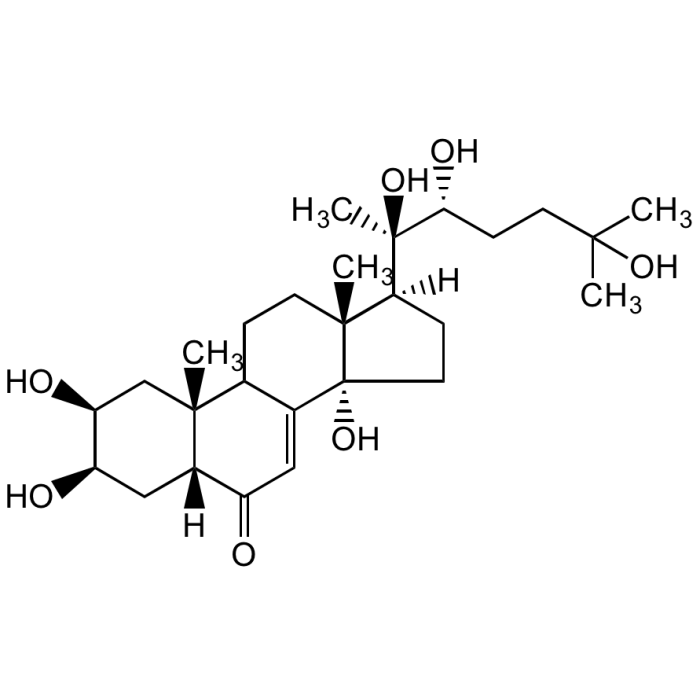
Ecdysterone
| Product Details | |
|---|---|
| Synonyms | 20-Hydroxyecdysone; β-Ecdysone; 2β,3β,14α,20β,22,25-Hexahydroxy-7-cholesten-6-one; Insect moulting hormone; Polypodine A |
| Product Type | Chemical |
| Properties | |
| Formula |
C27H44O7 |
| MW | 480.63 |
| CAS | 5289-74-7 |
| Source/Host Chemicals | Synthetic |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | White to off-white powder. |
| Solubility | Soluble in methanol (20mg/ml), DMSO or ethanol. |
| Identity | Determined by 1H-NMR. |
| Declaration | Manufactured by Chemodex. |
| Other Product Data |
Click here for Original Manufacturer Product Datasheet |
| InChi Key | NKDFYOWSKOHCCO-MQMWGCQISA-N |
| Smiles | [H][C@@]1(CC[C@@]2(O)C3=CC(=O)[C@]4([H])C[C@@H](O)[C@@H](O)C[C@]4(C)C3CC[C@]12C)[C@@](C)(O)[C@H](O)CCC(C)(C)O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Protect from light and moisture. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
A member of the ecdysteroid family. Ecdysone receptor (EcR) agonist. More potent than ecdysone. Induces the expression of genes coding for proteins that the larva requires, and it causes chromosome puffs (sites of high expression) to form in polytene chromosomes. Plays a key role in insect development, cell proliferaton, growth and apoptosis by controlling gene expression involved in moulting and metamorphosis. It acts through a heterodimeric receptor comprising the ecdysone receptor and the ultraspiracle proteins (USP). Used for controlled gene expression in scientific research, agriculture and medicine. Used for the development of selective insect growth regulators for use as environmentally benign insecticides. Shows biological effects on mammalian species, including antiepileptic, antidiabetic, antiobesity, ROS-inhibiting activity.ht produces vitamin D3. UV/Vis: λmax 271, 282, 293nm.
(1) F. Engelmann; Science 174, 1041 (1971) | (2) S. Tsujiyama, et al.; Jpn. J. Pharmacol. 68, 133 (1995) | (3) E.H. Baehrecke, et al.; Arch. Insect Biochem. Physiol. 33, 231 (1996) | (4) R. Hanaya, et al.; Jpn. J. Pharmacol. 74, 331 (1997) | (5) L.M. Riddiford, et al.; Vitam. Horm. 60, 1 (2000) | (6) C.S Thummel; Insect Biochem. Mol. Biol. 32, 113 (2002) | (7) L.D. Graham; Expert Opin. Biol. Ther. 2, 525 (2002) | (8) P. Kizelsztein, et al.; Am. J. Physiol. Endocrinol. Metab. 296, E433 (2009) | (9) J. Hu, et al.; J. Cell Biochem. 111, 1512 (2010)