Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
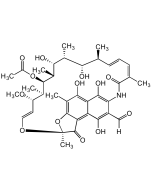
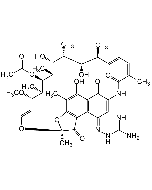
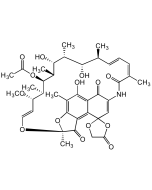
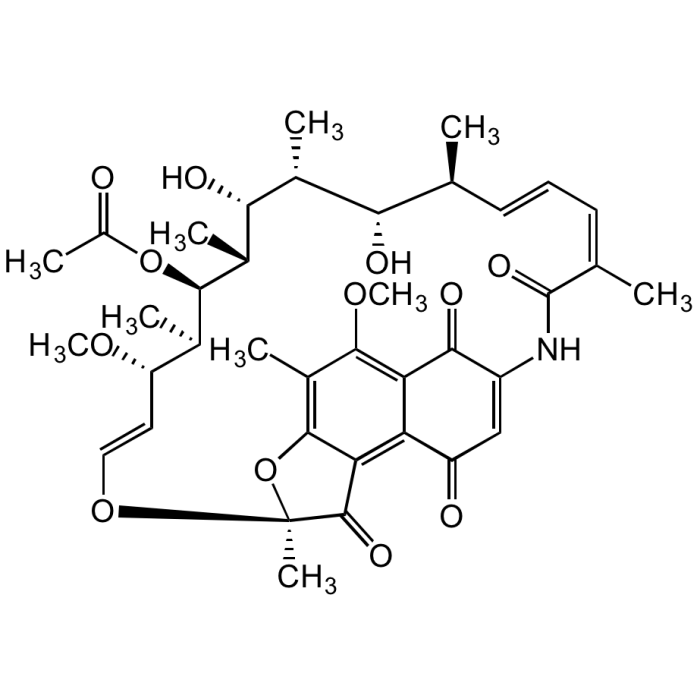
Rifamycin S, 8-Methyl-
As low as
150
CHF
CHF 150.00
In stock
Only %1 left
AG-CN2-0337-M01010 mgCHF 150.00
AG-CN2-0337-M05050 mgCHF 600.00
| Product Details | |
|---|---|
| Synonyms | 8-Methylrifamycin S; 8-O-Methylrifamycin S; 1,4-Dideoxy-1,4-dihydro-8-O-methyl-1,4-dioxo-rifamycin |
| Product Type | Chemical |
| Properties | |
| Formula |
C38H47NO12 |
| MW | 709.8 |
| CAS | 42898-13-5 |
| Source/Host Chemicals | Semisynthetic. |
| Purity Chemicals | ≥90% (HPLC) |
| Appearance | Yellow to orange powder. |
| Solubility | Soluble in DMSO, aqueous acetonitrile or ethanol. |
| Identity | Determined by 1H-NMR and MS. |
| InChi Key | CWRGYNMOWZQIMN-KSWODUNLSA-N |
| Smiles | O=C1C(NC(/C(C)=C\C=C\[C@H](C)[C@H](O)[C@@H](C)[C@H]([C@@H](C)[C@@H]([C@H](C)[C@H](/C=C/O2)OC)OC(C)=O)O)=O)=CC(C3=C4C(O[C@@]2(C)C4=O)=C(C)C(OC)=C31)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice |
Keep cool and dry. Protect from light. Protect from light when in solution. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Ansamycin antibiotic.
- Selective inhibitor of bacterial DNA-dependent RNA polymerase (RNAP).
- Effective against mycobacteria and therefore used in research of tuberculosis, leprosy and Mycobacterium avium complex (MAC) infections.
Product References
- The constitution and configuration of rifamycins B, O, S and SV: W. Oppolzer & V. Prelog; Helv. Chim. Acta 56, 2287 (1973)
- Letter: Electronegative groups at C-3 of rifamycin S enhance its activity toward DNA-dependent RNA polymerase: M.F. Dampier & H.W. Whitlock; JACS 97, 6254 (1975)