Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
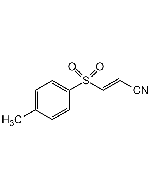
Dapansutrile
As low as
40
CHF
CHF 40.00
In stock
Only %1 left
AG-CR1-3535-M01010 mgCHF 40.00
AG-CR1-3535-M05050 mgCHF 150.00
AG-CR1-3535-M250250 mgCHF 525.00
| Product Details | |
|---|---|
| Synonyms | 3-(Methylsulfonyl)propanenitrile |
| Product Type | Chemical |
| Properties | |
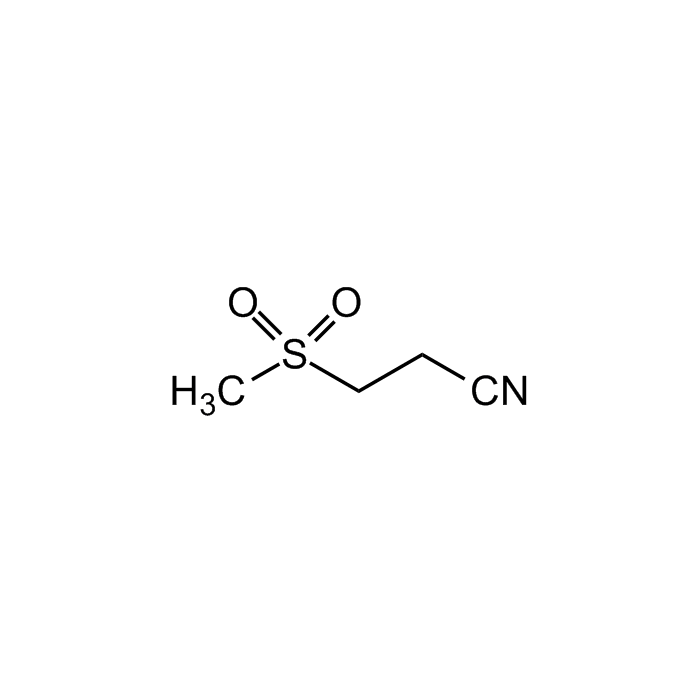
| Formula |
C4H7NO2S |
| MW | 133.2 |
| CAS | 54863-37-5 |
| Purity Chemicals | ≥98% (NMR) |
| Appearance | White to off-white solid. |
| Solubility | Soluble in DMSO (20mg/ml) or water (3mg/ml). |
| Identity | Determined by 1H-NMR. |
| InChi Key | LQFRYKBDZNPJSW-UHFFFAOYSA-N |
| Smiles | CS(CCC#N)(=O)=O |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
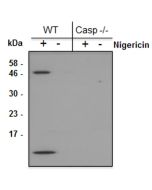
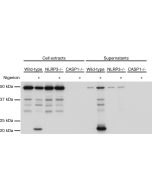
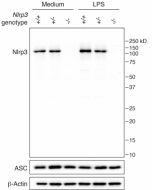
- Dapansutrile is a potent, selective and orally active inhibitor of the NLRP3 inflammasome. It directly binds to the ATP-binding motif of NLRP3 NACHT domain and inhibits NLRP3 adenosine triphosphatase (ATPase) activity, resulting in the suppression of NLRP3 inflammasome assembly (interaction between NLRP3, Asc and caspase-1) and activation.
- Dapansutrile is an anti-inflammatory agent by inhibiting the release of inflammatory signals such as IL-1β and IL-18. However, it does not impact the mRNA levels of the NLRP3, Asc, caspase-1, IL-1β and IL-18 genes. In vitro, nanomolar concentrations of Dapansutrile reduce IL-1β and IL-18 release following canonical and noncanonical NLRP3 inflammasome activation.
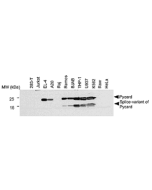
- Dapansutrile is selective for NLRP3 over NLRP4 and AIM2 inflammasomes at concentrations up to 100μM. Known other targets of Dapansutrile include several phosphorylated kinases such as Src, Fyn, HcK and STAT3.
- Dapansutrile has a preferable toxicological safety profile for in vivo studies. Dapansutrile reduces myeloperoxidase (MPO), CXCL1 and IL-6 levels in peritoneal fluid as well as IL-1β levels in liver, lung, spleen and skeletal muscle in a mouse model of LPS-induced systemic inflammation. It reduces joint swelling and markers of inflammation in synovial fluid in a mouse model of acute arthritis and displays cardioprotective effects in a mouse model of cardiac ischemic reperfusion injury and it rescues cognitive impairment in a mouse model of Alzheimer's disease.
- Dapansutrile reduces IL-1β release in blood monocytes isolated from patients with cryopyrin-associated periodic syndrome (CAPS), an autoinflammatory disorder characterized by gain-of-function mutations in NLRP3.
- Dapansutrile was tested in clinical trials and has been proposed as a beneficial compound for the remedy of osteoarthritis and gouty arthritis. In addition, preclinical research has proposed Dapansutrile to be potentially beneficial for heart failure and multiple sclerosis.
- Dapansutrile inhibits tumor-derived NLRP3 to reduce MDSCs expansion and to enhance antitumor immunity, resulting in reduced tumor growth. NLRP3 inhibition and anti-PD-1 treatment significantly increased the antitumor efficacy of the monotherapy by limiting MDSC-mediated T cell suppression and tumor progression.
- Dapansutrile prevented the loss of motor function, reduced the levels of α-synuclein, modulated pro-inflammatory markers in the nigrostriatal areas of the brain, and protected dopaminergic neurons from degeneration in the MPTP model of Parkinson’s disease (PD).
Product References
- OLT1177®, a β-sulfonyl nitrile compound, safe in humans, inhibits the NLRP3 inflammasome and reverses the metabolic cost of inflammation: C. Marchetti, et al.; PNAS 115, E1530 (2018)
- NLRP3 inflammasome inhibitor OLT1177® suppresses joint inflammation in murine models of acute arthritis: C. Marchetti, et al.; Arthritis Res. Ther. 20, 169 (2018)
- OLT1177 (Dapansutrile), a Selective NLRP3 Inflammasome Inhibitor, Ameliorates Experimental Autoimmune Encephalomyelitis Pathogenesis: A. Sanchez-Fernandez, et al.; Front. Immunol. 10, 2578 (2019)
- The NLRP3 inflammasome inhibitor, OLT1177® (Dapansutrile), reduces infarct size and preserves contractile function after ischemia reperfusion injury in the mouse: S. Toldo, et al.; J. Cardiovasc. Pharmacol. 73, 215 (2019)
- The NLRP3 inflammasome inhibitor OLT1177 rescues cognitive impairment in a mouse model of Alzheimer's disease: N. Lonnemann, et al.; PNAS 117, 32145 (2020)
- Phase 1B, Randomized, Double-Blinded, Dose Escalation, Single-Center, Repeat Dose Safety and Pharmacodynamics Study of the Oral NLRP3 Inhibitor Dapansutrile in Subjects With NYHA II-III Systolic Heart Failure: G.F. Wohlford, et al.; J. Cardiovasc. Pharmacol. 77, 49 (2020)
- Dapansutrile, an oral selective NLRP3 inflammasome inhibitor, for treatment of gout flares: an open-label, dose-adaptive, proof-of-concept, phase 2a trial: V. Kluck, et al.; Lancet Rheumatol. 2, e270 (2020)
- Pharmacological targeting of NLRP3 deubiquitination fortreatment of NLRP3-associated inflammatory diseases: G.-M. Ren, et al.; Sci. Immunol. 6, eabe2933 (2021)
- Targeting tumor-derived NLRP3 reduces melanoma progression by limiting MDSCs expansion: I.W. Tengesdal, et al.; PNAS 118, e2000915118 (2021)
- Pharmacologic inhibition of NLRP3 reduces the levels of α-synuclein and protects dopaminergic neurons in a model of Parkinson's disease: J. Amo-Aparicio, et al.; J. Neuroinflammation 20, 147 (2023)