Cookie Policy: This site uses cookies to improve your experience. You can find out more about our use of cookies in our Privacy Policy. By continuing to browse this site you agree to our use of cookies.
AdipoGen Life Sciences
MCC950 . sodium salt
As low as
40
CHF
CHF 40.00
In stock
Only %1 left
AG-CR1-3615-M0011 mgCHF 40.00
AG-CR1-3615-M0055 mgCHF 65.00
AG-CR1-3615-M01010 mgCHF 100.00
AG-CR1-3615-M05050 mgCHF 330.00
BULK available!
| Product Details | |
|---|---|
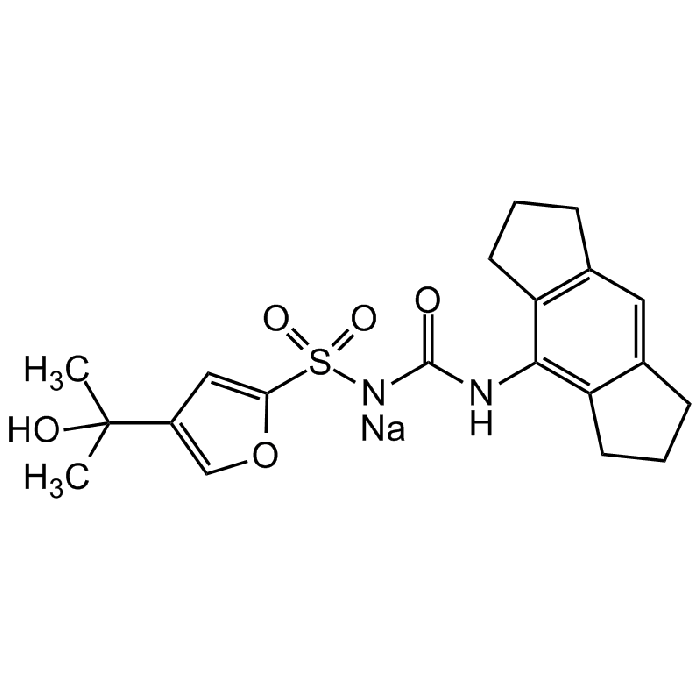
| Synonyms | MCC950; CP-456,773; CRID3; 1-(1,2,3,5,6,7-Hexahydro- s-indacen-4-yl)-3-[4-(1-hydroxy-1-methyl-ethyl)-furan-2-sulfonyl]urea |
| Product Type | Chemical |
| Properties | |
| Formula |
C20H23N2NaO5S |
| MW | 426.5 |
| CAS | 256373-96-3 |
| Purity Chemicals | ≥97% (HPLC) |
| Appearance | Off-white crystalline solid. |
| Solubility | Soluble in water (10mg/ml), DMSO (10mg/ml), dimethyl formamide or ethanol (1mg/ml). |
| Identity | Determined by 1H-NMR. |
| InChi Key | SNIGCXULUAXYGW-UHFFFAOYSA-N |
| Smiles | [NaH].CC(C)(O)C1=COC(=C1)S(=O)(=O)NC(=O)NC1=C2CCCC2=CC2=C1CCC2 |
| Shipping and Handling | |
| Shipping | AMBIENT |
| Short Term Storage | +4°C |
| Long Term Storage | -20°C |
| Handling Advice | Keep cool and dry. |
| Use/Stability | Stable for at least 2 years after receipt when stored at -20°C. |
| Documents | |
| MSDS |
 Download PDF Download PDF |
| Product Specification Sheet | |
| Datasheet |
 Download PDF Download PDF |
Description
- Potent, selective and orally available NLRP3 inflammasome inhibitor.
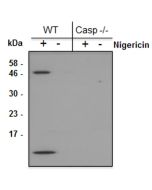
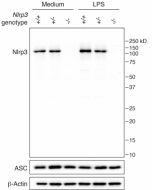
- Blocks the release of IL-1β in macrophages primed with LPS and activated with ATP or nigericin, but it does not inhibit NLRP1, NLRC4, AIM2, TLR2 signaling or priming of NLRP3.
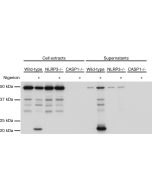
- Prevents oligomerization of ASC in cells stimulated with LPS and nigericin.
- Active in vivo, blocking the production of IL-1β and enhancing survival in mouse models of multiple sclerosis and cryopyrin-associated periodic syndrome (CAPS). Also active in ex vivo samples from individuals with Muckle-Wells syndrome.
- Potential therapeutic agent for NLRP3-associated syndromes, including autoinflammatory and autoimmune diseases.
Product References
- Glutathione s-transferase omega 1-1 is a target of cytokine release inhibitory drugs and may be responsible for their effect on interleukin-1beta posttranslational processing: R.E. Laliberte, et al.; J. Biol. Chem. 278, 16567 (2003)
- Novel synthesis of 1-(1,2,3,5,6,7-hexahydro-s-indacen-4-yl)-3-[4-(1-hydroxy-1-methylethyl)furan-2-sulfonyl]urea, an antiinflammatory agent: F.J. Urban, et al.; Synth. Commun. 33, 2029 (2003)
- The cytokine release inhibitory drug CRID3 targets ASC oligomerisation in the NLRP3 and AIM2 inflammasomes: R.C. Coll, et al.; PLoS One 6, e29539 (2011)
- A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases: R.C. Coll, et al.; Nat. Med. 21, 248 (2015)
- Taming the inflammasome: M. Levy, et al.; Nat. Med. 21, 213 (2015)
- The Nlrp3 inflammasome admits defeat: C.J. Gross & O. Gross; Trends Immunol. 36, 323 (2015)
- P2X7 receptor antagonism prevents IL-1β release from salivary epithelial cells and reduces inflammation in a mouse model of autoimmune exocrinopathy: M.G. Khalafalla, et al.; J. Biol. Chem. 292, 16626 (2017)
- Inhibition of the NOD-like receptor protein 3 inflammasome Is protective in juvenile influenza A virus infection: B.M. Coates, et al.; Front. Immunol. 8, 782 (2017)
- MCC950, the selective NLRP3 inflammasome inhibitor protects mice against traumatic brain injury: S. Ismael, et al.; J. Neurotrauma 35,1294 (2017)
- Inhibitory Effect and Mechanism of Arctium lappa Extract on NLRP3 Inflammasome Activation: Y.K. Kim, et al.; Evid. Compl. Altern. Med. 6346734 (2018)
- MCC950, a specific small molecule inhibitor of NLRP3 inflammasome attenuates colonic inflammation in spontaneous colitis mice: A.P. Perera, et al.; Sci. Rep. 8, 8618 (2018)
- α1-antitrypsin mitigates NLRP3-inflammasome activation in amyloid β1–42-stimulated murine astrocytes: T. Ebrahimi, et al.; J. Neuroinfl. 15, 282 (2018)
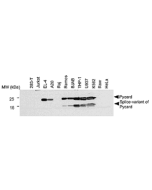
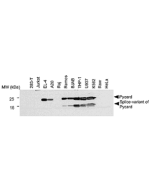
- Pyrin dephosphorylation is sufficient to trigger inflammasome activation in familial Mediterranean fever patients: F. Magnotti, et al.; EMBO Mol. Med. 11, e10547 (2019)
- Toxoplasma gondii activates a Syk-CARD9-NF-κB signaling axis and gasdermin D-independent release of IL-1β during infection of primary human monocytes: W.J. Pandori, et al.; PLoS Pathog. 15, e1007923 (2019)
- Interleukin-1β suppression dampens inflammatory leukocyte production and uptake in atherosclerosis: J. Hettwer, et al.; Cardiovasc. Res. 117, 2778 (2022)
- Acute suppression of mitochondrial ATP production prevents apoptosis and provides an essential signal for NLRP3 inflammasome activation: B.S. Saller, et al.; Immunity 58, 90 (2025)